Abstract
Introduction:
Multiple myeloma (MM) is caused by heterogeneous genomic alterations that lead to activation of the MYC oncogenic pathway (Larrayoz et al., 2023). Moreover, MYC activity plays a role in immune escape by MM cells and alteration of the bone marrow microenvironment as the disease progresses, which in turn affects response to immunotherapy. Downstream targets and pathways of MYC, such as polyamine (PA) pathway, are an important avenue for therapeutic anticancer interventions for targeting MYC that is generally considered to be “undruggable”. In the present study, we therefore evaluated the effect of two polyamine inhibitors, ivospemin and difluoromethylornithine (DFMO), on growth and viability of myeloma cell lines. SBP-101, an analogue of the naturally occurring PA, spermine, is a polyamine metabolic inhibitor (PMI) that reduces native PA pools by inhibiting key synthetic enzymes and induction of catabolic enzymes. DFMO is an irreversible inhibitor of the ornithine decarboxylase and polyamine synthesis.
Methods:
Four myeloma cell lines: NCI-H929, RPMI-8226, ARP-1, and MM.1S, were exposed to varying concentrations of two PA inhibitors, SBP-101 and DFMO, based on the data available from clinical trials conducted for other malignancies. We analyzed the effects of these treatments on cell proliferation, cell viability, and the induction of apoptosis. The analyses were done separately for each compound as well as their combination. To assess cytotoxicity and cell proliferation, we used the Incucyte® SX5 Live-Cell Analysis System. Additionally, the detection of apoptosis was performed using Annexin V and propidium iodide flow cytometric assay.
Results:
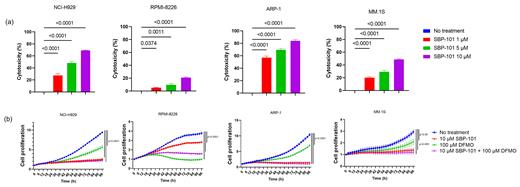
Exposure of the MM cell lines to Ivospemin at increasing concentrations ranging from 1µM to 10 µM showed a significant decrease in cellular proliferation in all four MM cell lines. The mean percentage of cytotoxicity observed at 10 µM concentrations of Ivospemin in NCI-H929, RPMI-8226, ARP-1, and MM.1S cell lines were 69.17, 20.75, 83.92, and 48.71 respectively (Fig. 1a). Similarly, when we exposed the MM cell lines to DFMO at increasing concentrations ranging from 1 µM to 100 µM, we also saw a significant decrease in cellular proliferation in all four cell lines, with the most pronounced effect observed in RPMI-8226. The percentage of cells killed at DFMO 10 µM in NCI-H929, RPMI-8226, ARP-1, and MM.1S cell lines were 32.67, 63.61, 28.24, and 16.06 respectively.
Moreover, when we combined Ivospemin at 10 µM and DFMO at 100 µM, we observed a complete abolition of cell growth in all the cell lines (Fig. 1b). Additionally, the treatment of the myeloma cell lines with either Ivospemin, DFMO alone, or in combination, showed an induction of apoptosis, as revealed by flow cytometric analysis (data not shown). These findings indicate that both Ivospemin and DFMO have significant anti-proliferative effects on MM cell lines, and their combination leads to even more potent growth inhibition and induction of apoptosis.
Conclusion: The polyamine inhibitors,Ivospemin and DFMO, exhibited a marked anti-proliferative effect in the four human MM cell lines studied when used alone or in combination. These results confirm the anti-neoplastic potential of SBP-101 and DFMO and offer a compelling rationale for its clinical development as a potentially promising treatment option for multiple myeloma.
Fig. 1. Effect of varying concentration of Ivospemin (SBP-101) treatment on myeloma cell lines (a). The data is represented as mean ± SEM of three replicates. A combination of both Ivospemin (SBP-101) and DFMO improved the cytotoxicity as compared to when used alone (b).
Disclosures
Liu:N/A: Patents & Royalties: Related to cell therapy and the safety switch described (intellectual property). Patchva:N/A: Patents & Royalties: Related to the safety switch described (intellectual property). Bruckheimer:Panbela Theraputics: Current Employment. Neelapu:Merck: Consultancy, Other: Advisory Board Member; Sellas Life Sciences: Consultancy, Other: Advisory board member; Kite, A Gilead Company: Consultancy, Other: Advisory Board Member, Research Funding; Allogene: Consultancy, Other: Advisory board member, Research Funding; Athenex: Consultancy, Other: Advisory board member; Synthekine: Consultancy, Other: Advisory board member; Incyte: Consultancy, Other: Advisory board member; Adicet Bio: Consultancy, Other: Advisory board member, Research Funding; Bristol Myers Squibb: Consultancy, Other: Advisory Board Member, Research Funding; Bluebird Bio: Consultancy, Other: Advisory board member; Fosun Kite: Consultancy, Other: Advisory board member; Sana Biotechnology: Consultancy, Other: Advisory board member, Research Funding; Caribou: Consultancy, Other: Advisory board member; Astellas Pharma: Consultancy, Other: Advisory board member; Morphosys: Consultancy, Other: Advisory board member; Janssen: Consultancy, Other: Advisory board member; Chimagen: Consultancy, Other: Advisory board member; Immunoadoptive Cell Therapy Private Limited: Consultancy, Other: Scientific Advisory Board; Orna Therapeutics: Consultancy, Other: Advisory board member; Takeda: Consultancy, Other: Advisory board member; Carsgen: Consultancy; Precision Biosciences: Research Funding; Longbow Immunotherapy: Current holder of stock options in a privately-held company; N/A: Patents & Royalties: Related to cell therapy and the safety switch described (intellectual property). Saini:GSK: Research Funding; Panbela Theraputics: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal